Metal quality does matter when you are focusing on building something to last long, and at the same time, it can handle the wear and tear of life.
Quality does matter to people; sacrificing quality will lead to a wrong product.
The unique properties behind the metal make them more interesting than others.
In the market, you will find different varieties of metals, each with its own perks.
The quality of the metals will be various, some might offer flexibility while others will be much harder than others, and at the same time, you will also encounter soft metals.

In your mind, you are probably wondering why anyone would need a soft metal for anything.
In a way, you are right; if you ever see any structure or object made of metal, the first thing that comes to your mind is that the item must be heavy.
As our mind quickly considers any form of metal as a heavier object. But soft metals do exist;
they have a variety of uses; some of them are used in making jewellery, and some do find their way to the construction environment.
You will find soft metal easily in the market, but when the question turns to what is the most delicate metal, it will be complicated for anyone to find the proper answer.
This article will explore the righteous contender for the title of softest metal in the world. We are going to focus on Caesium.
What is the Softest Metal?
When you see the word soft in a metal world, it refers to the metal’s malleability.
The metal can be quickly hammered into the paper or sheet. Soft metals can also be easily cut with a knife.
As we mentioned earlier, soft metals are used for jewellery purposes, so some of the best examples of soft metals you have in your household are gold, silver, lead, and copper.
Some may not know aluminium is also seen as a soft metal, but it is not as malleable as others.
So how do people conclude to find the softest metal in the market? Since all of them share the same properties, it can be difficult for anyone to find the exact properties that make the metal Soft.
For proper analysis, the Mohs hardness scale identifies the softness of various metals.
Caesium gets used in the industry as a catalysis promoter. Its job is to boost the performance of other metal oxides in the capacity and hydrogenation of other organic compounds.
Metal is also used to make optical glass. Over the years, Caesium found a way into various applications of work.
Currently, Caesium is used to remove traces of oxygen from light bulbs and vacuum tubes.

Introduction of Caesium
Of all the Caesium elements, Caesium has the lowest ionization potential and is also non-radioactive; gallium, francium, rubidium, mercury, and Caesium are the only pure metals that can be liquid at room temperature.
Ti reacts to cold water and ice explosively. The temperature here should stay above the -177 threshold degree in Fahrenheit.
Bernic Lake Manitoba has estimated that around 300k tons of polluted ore exist within the lakebed averaging 20% Caesium by weight.
Caesium has approximately 39 isotopes, which makes it the second most known isotope of all the elements, second to francium.
The atomic masses of these isotopes range from 112 to 151.
Out of all the stops, only Cs-133 can be perceived as naturally stable.
All other isotopes have half-lives ranging from a few days to a few seconds or less—cs-137 proceeds in nuclear power plants and atomic detonation of weapons.
In 1986, a large quantity of Cs-137 was released into the atmosphere.
Explosive Aspect
Caesium is one of the heaviest alkali metals and is the most reactive. It is highly explosive when it comes into contact with water.
The content generates hydrogen gas which is then heated by thermal energy released simultaneously.
This causes the ignition of the gas, and an explosion soon follows. The reactive component of Caesium is what makes ice trigger a large explosion.
Caesium can also ignite the air because of Caesium hydroxide and other various oxides.
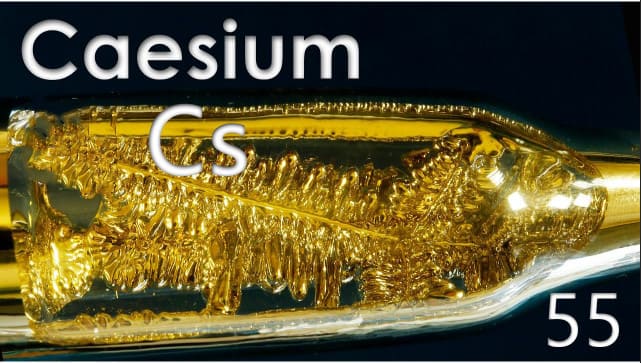
Caesium Inclusion in Everyday Lives
The mild toxic component of Caesium is hazardous, but it never comes in contact with humans.
The presence of potassium is what makes Caesium a toxic element. Adding to the diet is the last thing you want to do with Caesium, a study that focused on lab rats showed all the rats that had Caesium died.
This shows Cs are not effectively accumulated in the body; some of them, for example, Cs-134 and Cs-137, will cause radioactivity problems depending on the area.
Properties of Caesium Breakdown
Here we will look at Caesium and break down almost all the details to help you know the metal and all of the essential features that helped it earn the title of softest metal in the world.
We will focus on uses, appearance, biological role, history, and more to uncover the most basic details about the metal to the front.
Caesium is the softest metal in the world. The metal here is mild, silvery god, and malleable. It is seen as the most electropositive and can react explosively with cold water. The metal reacts to the ice at a temperature above -116*c.
Scientific Breakdown of Caesium
| Atomic number | 55 |
| Atomic mass | 132.9054 g.mol -1 |
| Electronegativity according to Pauling | 0.7 |
| Melting point | 28.5°C, 83.3°F, 301.7 K |
| Period | 6 |
| Boiling point | 671°C, 1240°F, 944 K |
| Density | 1.9 g.cm3 at 20°C |
| Melting point | 28.4 °C |
| Relative atomic mass | 132.905 |
| Boiling point | 669 °C |
| Van Der Waals radius | 0.267 nm |
| Ionic radius | 0.167 |
| CAS number | 7440-46-2 |
| Key isotopes | 133Cs |
| Isotopes | 12 |
| Electronic shell | [ Xe ] 6s1 |
| The energy of the first ionization | 375.6 kJ.mol -1 |
| Discovered by Fustov Kirchhoff in | 1860 |
[Ref – Link]
History behind Caesium
The traces of history with Caesium date back to the year 1800, specifically 1846, by a man named Carl Plattner.
He investigated the mineral pollucite or as known as Caesium aluminium silicate. His investigation stopped at 93%, as he can only account for that number of the elements I t contained.
He quickly ran out of material to analyze. Later it was revealed he mistook Caesium for sodium and potassium.
Then move forward to the year 1860, where a man named Gustav Kirchhoff and Robert Bunsen in Germany discovered the existence of Caesium.
They examined the mineral from Durkheim and saw the lines in the spectrum they never saw, which means that an element has risen to the surface.
In the analysis, they managed to produce 7 grams of Caesium chloride from the source but failed to create a new sample of the metal.
That particular aspect of the investigation goes to Carl Theodor Setterberge at the University of Bonn, who got electrolysis of molten Caesium cyanide CsCN.

Caesium Appearance
Caesium is a soft, gold-coloured material that can react to water and air. It can get quickly attacked by air and reacts explosively in water.
Caesium Uses
As we have mentioned before, the most common usage of Caesium is used as drilling fluid. Some do make it to other optical glass industries as catalyst promoters.
Vacuum tubes and radiation monitoring equipment do have Caesium in them.
The modern market has found various uses for Caesium, and now you will see them used in drilling fluid for the oil industry.
Caesium’s stable isotopic nature makes it environmentally safer than other metal compounds used for oil drilling. Caesium can be found in atomic clocks for accurate measurement of time.
Other uses for Caesium included hydrogenation, medical treatment of cancer, organic chemistry, molecular biology, oxidation, and pyrotechnics for infrared flares, spectrophotometer, exhaust reduction of military aircraft, industrial applications for gauges and devices n in many magnetometers.
The prominent use of Caesium lies in the Caesium clock or atomic clock. These clocks are used in the internet and mobile phone network; some make it a Global positioning system or GPS satellites.
The electron resonance frequency of the Caesium atom is 9,192,631,770/ sec. Some Caesium cloaks are accurate to 1 second in 15 million years.
Caesium Atomic Data
Here is the detailed list of all the atomic data present in Caesium.
| 1. Atomic radius, non-bonded (Å) | 3.43 |
| 2. Covalent radius (Å) | 2.38 |
| 3. Electron affinity (kJ mol−1) | 45.505 |
| 4. Electronegativity (Pauling scale) | 0.79 |
| 5. Ionization energies (kJ mol−1) | |
| 1st – | 375.705 |
| 2nd- | 2234.353 |
| 3rd – | |
| 4th – | |
| 5th – | |
| 6th – | |
| 7th – | |
| 8th – |
[Ref – Link]
Caesium Oxidation states and isotopes
This section covers the oxidation states and isotopes of Caesium.
| Common oxidation states | 1 |
| Isotopes | |
| Isotope | 133Cs |
| Atomic mass | 132.905 |
| Natural abundance (%) | 100 |
| Half-life – | |
| Mode of decay – |
[Ref – Link]
Caesium supply risk
Here we are going to cover the supply risk Caesium.
| Relative supply risk | Unknown |
| Crustal abundance (ppm) | 3 |
| Recycling rate (%) | Unknown |
| Substitutability | Unknown |
| Production concentration (%) | Unknown |
| Reserve distribution (%) | Unknown |
| Top 3 producers | Unknown |
| Top 3 reserve holders | Unknown |
| The political stability of top producer | Unknown |
| The political stability of top reserve holder | Unknown |
[Ref – Link]
Advanced pressure and temperature data
| Specific heat capacity (J kg−1 K−1) | 242 |
| Young’s modulus (GPa) | Unknown |
| Shear modulus (GPa) | Unknown |
| Bulk modulus (GPa) | 1.6 |
[Ref – Link]
Vapour pressure [Ref – Link]
| Temperature (K) | Pressure (Pa) |
| 400 | 0.394 |
| 600 | – |
| 800 | – |
| 1000 | – |
| 1200 | -Baro |
| 1400 | – |
| 1800 | – |
| 2000 | – |
| 2200 | – |
| 2400 | – |
Other Soft Metals
Other soft metal lists include lead, mercury, Lithium, and Aluminum. Most of them enter the temperate zone, but they are not considered the most delicate metal.
But most of the perks from Caesium adapt well with other soft metals on the list. Sometimes, you will encounter a different form of metal, lead is a weighty metal, but it is soft and malleable.
Lead
Lead has a melting point and can be cut with a knife. We see lead used in batteries, ammunition, pipes, and solder.
Despite its softness, it can be very poisonous, and exposure to information can cause damage to the brain and nervous system.
Mercury
Mercury is soft but doesn’t obtain the title of the most delicate metal. The mercury metal makes its way to thermometers, barometers, and fluorescent light bulbs.
It is very poisonous and causes a massive range of health problems. Mercury exposure will lead to brain, kidney, and lung damage.
At the same time, mercury posting includes headaches, dizziness, nausea, and skin rashes.
Lithium
Lithium is the lightest metal on the table; it is a rare metal, as it reacts with water and air to form lithium hydroxide and lithium oxide.
The reactions can be dangerous, and people handle lithium with care.
Aluminum
One of the most abundant metals on the planet, it is used in cans, foil, and various kitchen appliances and utensils.
It is very reactive, and it reacts to oxygen to form aluminium oxide; the reaction is called oxidation, which can cause corrosion, weakening the Aluminum and making it brittle.
Conclusion
While the title of the softest metal ever goes to the Caesium. All the other contenders, such as Aluminum, lead, mercury, lithium, and lead, are also seen as soft, but they are very dangerous if not handled adequately with guidance or care.
FAQ
Here are some of the most asked questions about Caesium on the internet.
Where do they find Caesium in the world?
Commonly they are found in the minerals pollucite and lepidolite. Pollucite is found in large quantities at Bernic Lake, Manitoba.
The preparation of the element goes on in countries such as Canada and USA. But the most commercial production is lithium production.
Which soft metal is most reactive?
That title will go to Caesium, as it is the most reactive of all the elements; that is why Caesium is stored in an airtight container and prevents it from oxidizing.
When was Caesium discovered?
The first sign of Caesium was discovered in the year 1846.
Which soft metal is used in jewellery?
Lead and tin are used in the jewellery crafting process. Lead is a very dense metal, which is why it is used for heavy-duty applications like X-ray shielding.
On the other hand, you have tin, often used in plating and coating other metals and giving them a shiny finish.
Can soft metals be cut with a knife?
Yes, soft metals can be cut through knives and are very reactionary to outside temperatures and elements.
They are usually stored and handled by professionals with experience who know how to handle the situations without making them worse.

 (+86) 188-2253-7569
(+86) 188-2253-7569



